| |
You are here: MIT OpenLabWare » Protein Caging
» Module Home
Module Home
Semisynthesis of unnatural amino acid mutants of paxillin
B Imperiali
E M Vogel
Research question: Can we synthesize a photolabile precursor to the cellular
migration protein paxillin and characterize it in vivo?
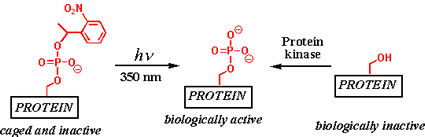
Paxillin is a complex, multi-domain phosphoprotein that coordinates
the cell's ability to move
and communicate. MIT Professor of Chemistry and Biology Barbara Imperiali and her graduate student,
Elizabeth Vogel, developed a method to semisynthesize a precursor of paxillin that can be activated
using a pulse of light (as shown in the image above). This tool enables biologists to understand the
role of a single phosphorylation event in the big picture of cell migration, which will provide
insight into the mechanisms of cancer proliferation and wound repair. They reported their results in
the March 2007 issue of Protein Science.
This module contains:
- An overview that contextualizes the research while providing some
chemical and biological
background.
- A detailed experiment timeline. Selected experiments from Elizabeth Vogel's six lab notebooks are annotated and presented in a timeline. Every experiment is linked to
scans of the corresponding pages in Elizabeth Vogel's lab notebook (PDF). Some experiments are linked to audiovisual supplementary information.
- Video interviews with Barbara Imperiali and Elizabeth Vogel. The
authors describe their career path, explain their research philosophies, and provide advice to
aspiring scientists. Barbara Imperiali also shares some of her travel pictures.
- Author profiles for Barbara Imperiali and Elizabeth Vogel (click on the links at the top of
this page).
- The full manuscript (HTML and PDF).
- A glossary of some obscure terms. The glossary is also accessible
from the Read the Paper page. Move the mouse over any hyperlinked word and its
definition will appear
in a tooltip.
| |